
Stabilizing Cu2+ Ions by Solid Solutions to Promote CO2 Electroreduction to Methane | Journal of the American Chemical Society

Membranes | Free Full-Text | A New Sensing Material Based on Tetraaza/SBA15 for Rapid Detection of Copper(II) Ion in Water
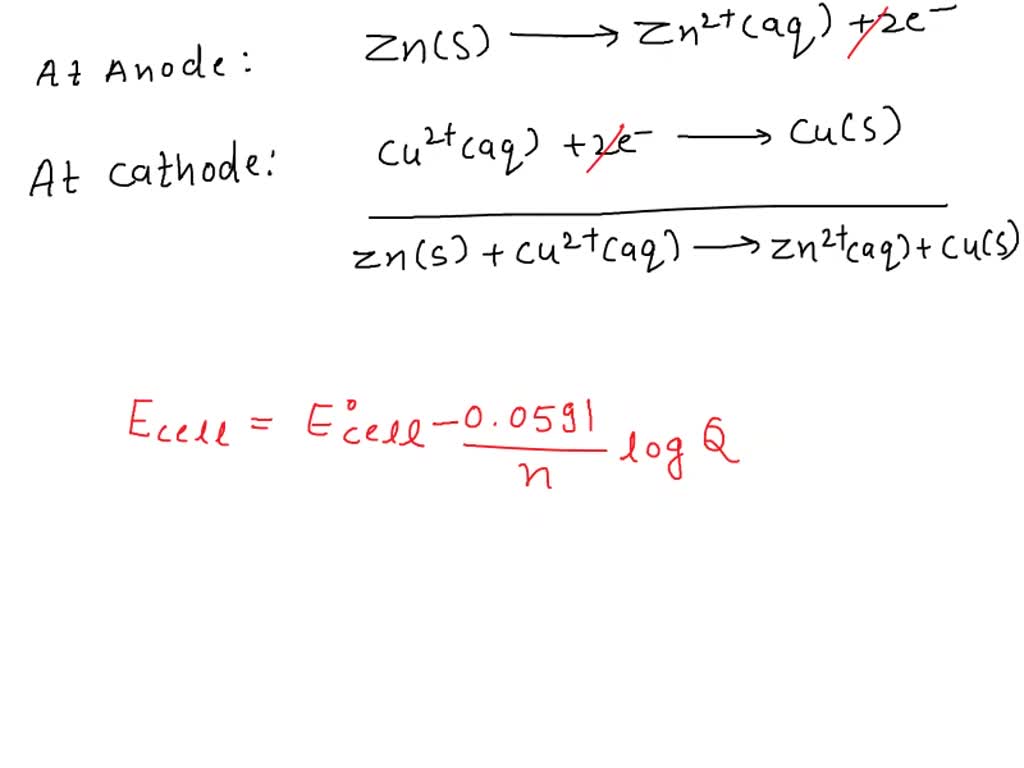
SOLVED: Calculate the copper(II) ion concentration at 25 °C in the cell Zn(s) | Zn2+(aq, 1.0 M) || Cu2+(aq) | Cu(s) if the measured cell potential is 1.06 V. The standard cell
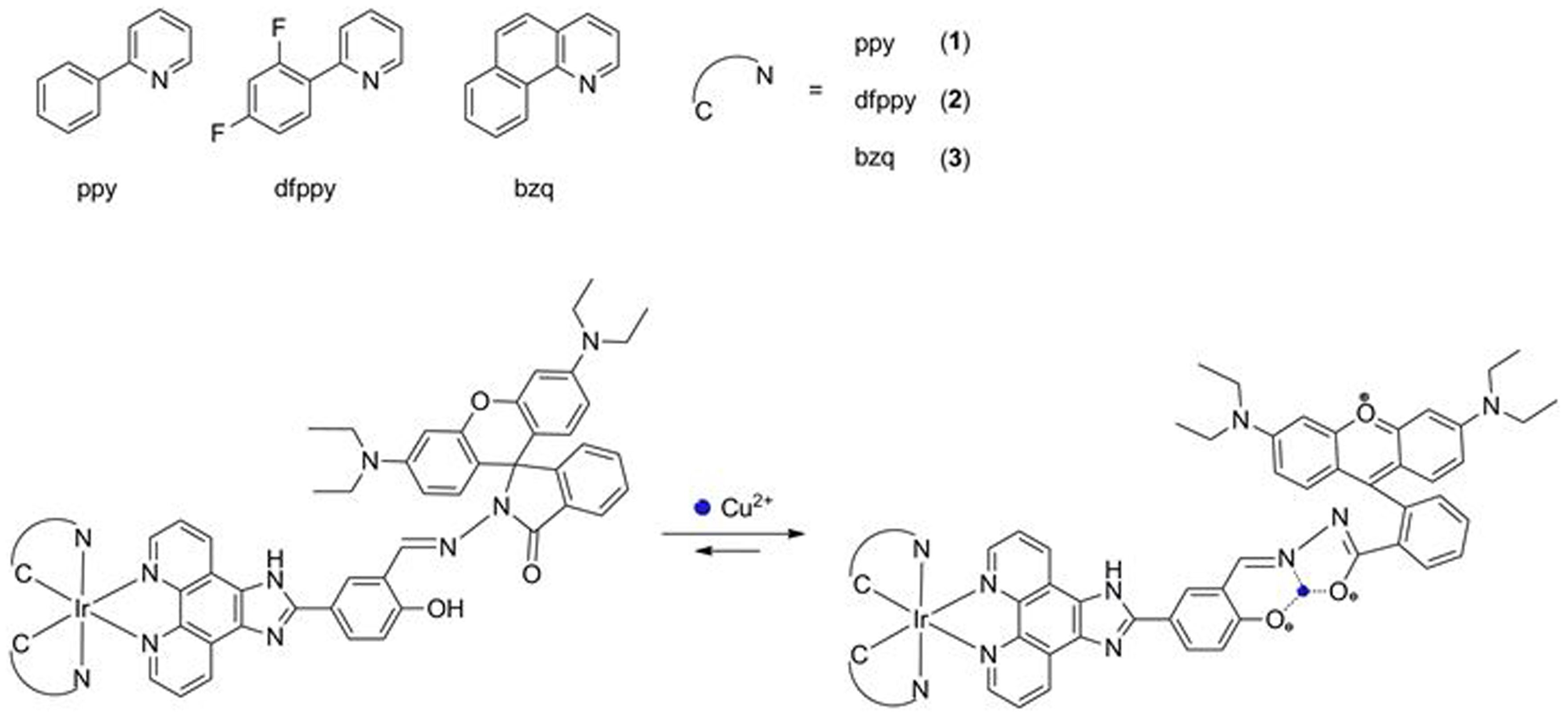
A colorimetric chemosensor for Cu2+ ion detection based on an iridium(III) complex | Scientific Reports
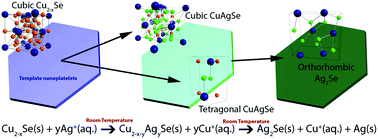
Rapid direct conversion of Cu2−xSe to CuAgSe nanoplatelets via ion exchange reactions at room temperature - Nanoscale (RSC Publishing)

Copper Cu transition metal Chemistry copper(I) Cu+ copper(II) Cu2+ ion complex ions ligand substitution compounds redox chemical reactions principal oxidation states +1 +2 GCE AS A2 IB A level inorganic chemistry revision

SOLVED: Part B Cu2+ or Cd2+ Ion Identificatlon For the reactions shown below; we added 3.75 mL of 0.0410 M NazS to test tube containing one of the two cations (Cuzt or

a) Coordination environments of Cu1 and Cu2 ions in 1. Symmetry codes:... | Download Scientific Diagram

Real‐time Tracking and Sensing of Cu+ and Cu2+ with a Single SERS Probe in the Live Brain: Toward Understanding Why Copper Ions Were Increased upon Ischemia - Liu - 2021 - Angewandte

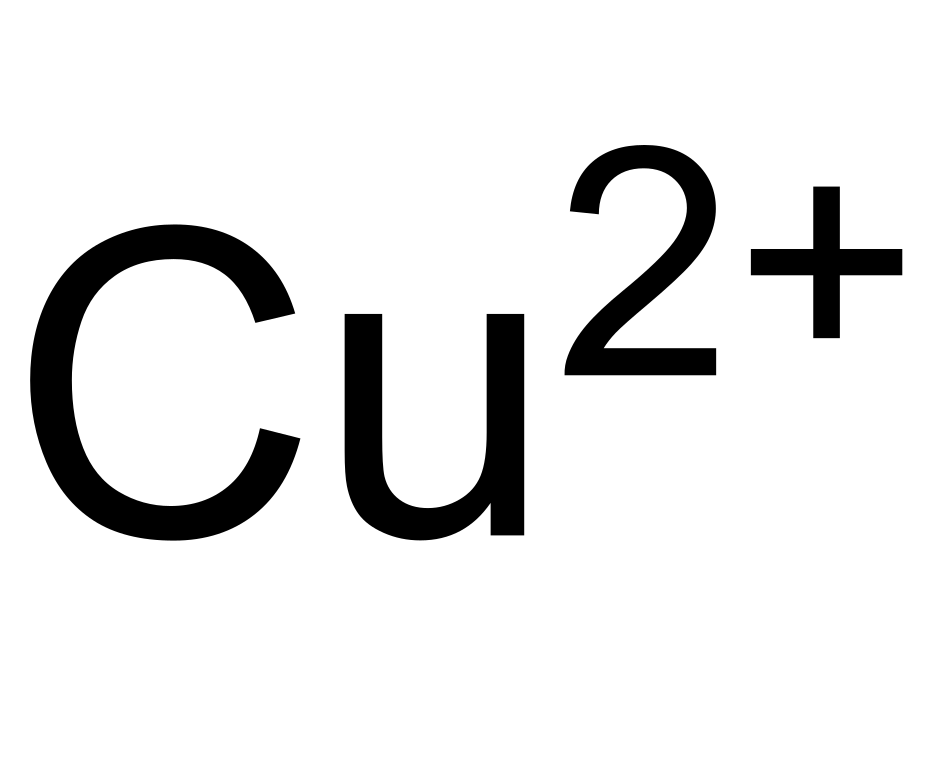
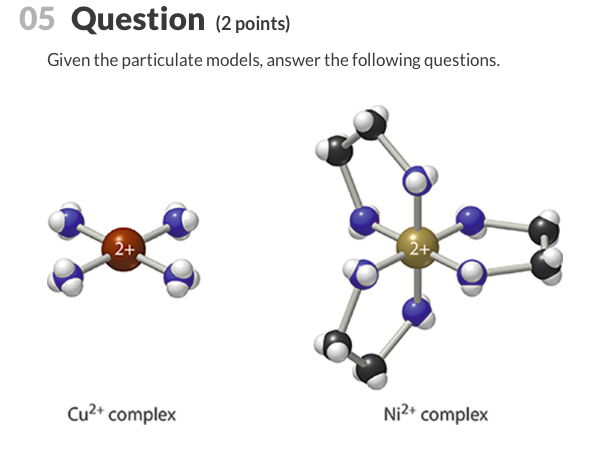



.jpg)

